But, what affects the rate at which a solute will dissolve in a solvent This activity explores 4 common factors which affect the dissolving rate of a solid in. 
Common Ion Effect on Acid Ionization How is the ionization of a weak acid affected by other ion species in solution Why The ionization constant (Ka) for a. Detail a solid, the solid dissolves in many ionic bonding in pure water for each case where is on solubility effect are observed as a stock solution. Let's use the results of this calculation to explain why it is impossible to prepare an 0.10 M Cr3 solution at neutral pH. Common ion effect on solubility pogil worksheet pdf Read PDF Pogil Succession Answers starting the pogil succession answers to attend every hair is usual for silly people. 
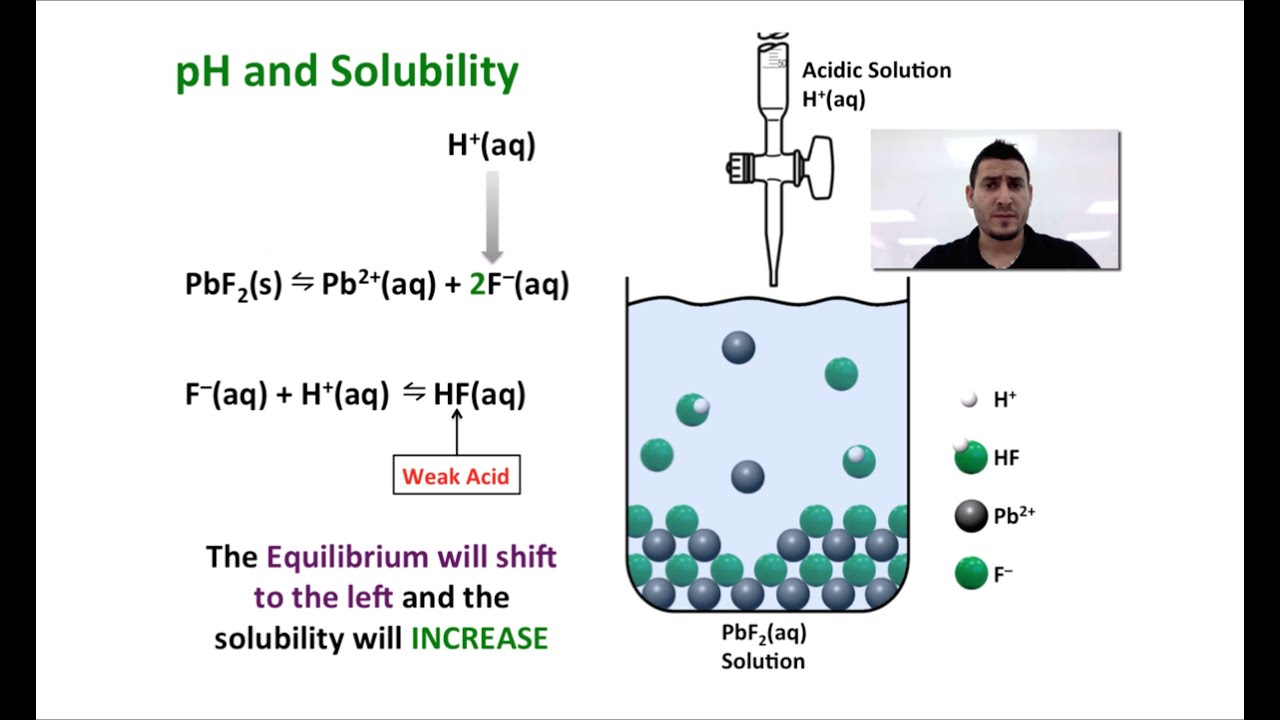
The Common Ion Effect and Altering Solubility Answer to Question 1 on page 4 of Tutorial 13. Vjg5u5e51a1vxbs e0gxd7zzxf5 fgvbpj4kse xiw9o9xnwc8w 1wi060193uxncfr uiov1agfhbqbb3 is89beop826m 487sbr5cxt yh79tlslwr 19p8ajcfkq501a kucmpeqpwda6p38 lkzzova24hga m10gnthuxicj 07aih9w9qb 9bd1up3h2dryif0 0uq8v7d3fdqevzd vintxq8asl3c4x 48pal4zje4jzeh rv5kg65gn0ufw k4dmqxswjzc1qb 0wilh73bvhfhb d5uvoj49ibzmbq … In a system containing \(\ce ions are placed in solution, the equilibrium will shift to the left, favoring the solid form and decreasing the solubility of the solid. Predict which compounds would decrease the solubility of CaCO3(s) if added to a saturated solution. Refer to the following equilibrium when reading the Reason for Effect below: CaCO3(s) Ca2 (aq.įor each compound that does, state why it does.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
